Explore Pepper Cloud
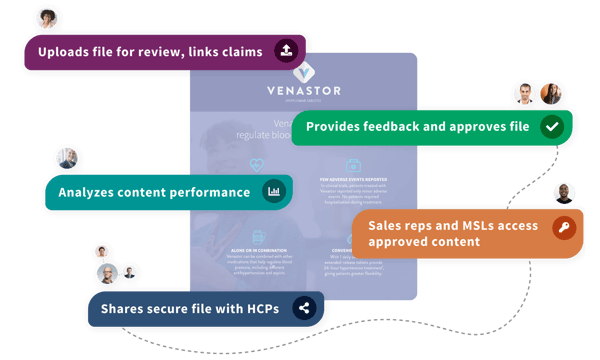
Pepper Cloud empowers life science companies to bring effective content to market faster through one connected platform of content review, sales and MSL enablement, and analytics software. With Pepper Cloud, teams accelerate their product commercialization through streamlined processes, improved stakeholder engagement, and access to dedicated industry experts.
Learn about Pepper Cloud
“Vodori is one of the best partners I have worked with and this was one of the best teams I have been a part of. Thank you.”
Senior Manager Fortune 500 Pharmaceuticals Company












.png?width=170&height=57&name=sight-sciences-logo@2x%20(1).png)