- Platform
- Platform Overview
- Life science compliance
- Enterprise-grade security
- Integrations
- Partner Ecosystem
- Workflow management
- Claims & evidence management
- Digital asset management
- Web annotation
- Health authority submission support
- Portals
- Cross channel publishing
- Global commercialization
- Vodori 360
- Reporting & analytics
- Solutions
- Resources
- Company
- Trust
Text recognition to suggest reference links, significantly reducing job preparation time
DOWNLOAD PDFChicago, IL – June 12, 2018 — Vodori, Inc. today announced a significant enhancement to Pepper Flow®, Vodori’s promotional review and approval system built exclusively for life sciences. Pepper Flow enables life science companies to review, collaborate, approve, and distribute content efficiently and compliantly.
During medical, legal, and regulatory (MLR) reviews, healthcare companies face tremendous scrutiny to clearly substantiate all claims made in their promotional materials. Preparers of those materials must therefore show the relationship between each claim and its substantiating literature or journal reference. In most promotional review systems, that relationship must be manually linked each time, even if the same claim is made in another promotional piece.
Today Vodori announced smart reference linking, the ability for Pepper Flow to automatically search for and suggest references based on previous usage in the application, drastically reducing the amount of time necessary to prepare a document for MLR review, while also ensuring all claims are appropriately substantiated.
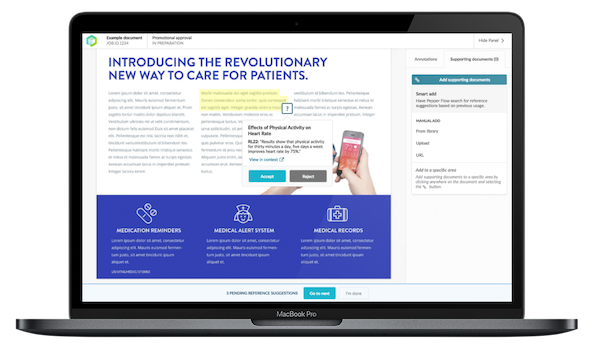
“This innovation builds on our desire to make job preparation faster, easier and more accurate. We’re excited to deliver a feature that can save our customers hundreds of work-hours each year. The tedium of re-linking previously used claims to reference pieces is virtually eliminated and our customers and their marketing agencies are excited to offload this work to Pepper Flow,” said Scott Rovegno, President and CEO of Vodori.
In addition to suggesting reference claims, customers can also see which promotional materials use each claim in the system, enabling a streamlined process for updating materials when a claim is deemed out of date or labeling information is revised.
Smart reference linking will be available to all customers in October 2018. To learn more, visit www.vodori.com/pepper-flow or contact Vodori at sales@vodori.com to see a full demo.
About Vodori
Since 2005, Vodori has been helping clients in the life science industry market, sell, support and service their clients. Vodori builds, implements, and supports Pepper Flow for its customers. Pepper Flow is part of the Pepper Cloud® Product Suite, a hub of SaaS products that enables organizations to manage, approve, publish, and withdraw content on a global scale, all from one platform.
Contact
Niki Drelicharz
Vodori, Inc.
312.940.7921
nicole.drelicharz@vodori.com
Other posts you might be interested in
View All Posts
3 min read
|
Latest Release of Pepper Flow® Promotional Review Software Announced | Vodori
Read More
3 min read
|
Vodori Announces the Pepper Flow Acceleration Program | Vodori
Read More
4 min read
|
DiaSorin Selects Vodori’s Pepper Flow to Support Rapid Growth and Increased Product Launches | Vodori
Read MoreSubscribe to Vodori
We'll send you content and resources on MLR best practices, collaboration tips, and more. By subscribing, you consent to allow Vodori to store and process the personal information submitted to receive communications from Vodori. You may unsubscribe at any time. For more information, review our Privacy Policy.


